
PRECISION IN MOLECULAR SYNTHESIS.
Precision synthesis of research compounds. Engineered, validated, and dispatched from ZQX laboratories.
Core Operations
[ Structural Overview ]
Synthesis
Custom molecular engineering executed under strict atmospheric controls. High-purity structural formulation protocols.
Verification Pipeline
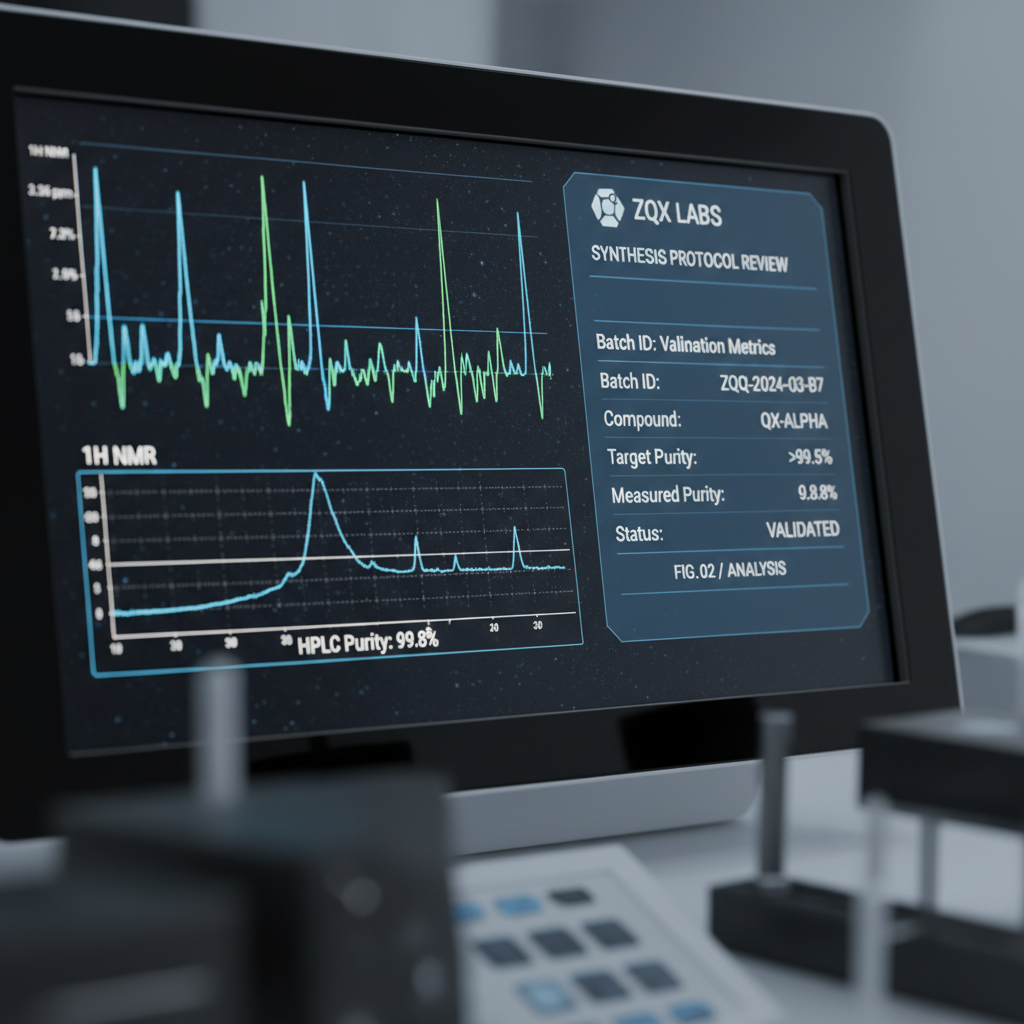
Internal analysis protocols utilizing advanced spectroscopy. Objective data yields and batch-specific validation.
Logistics Infrastructure
Direct fulfillment from ZQX synthesis facilities under climate-controlled transit calibrated for compound stability. Each shipment is sealed, logged, and dispatched within validated thermal parameters.

Custom Molecular Engineering
Every compound is manufactured at ZQX laboratories. Synthesis protocols execute on-site under the direct supervision of our research staff, governed by closed-chain procedural standards from precursor intake to final isolation.
Each stage proceeds within ZQX-operated facilities — raw precursor, intermediate, crystalline output. Structural integrity is held absolute through batch-specific validation, with redundant atmospheric and thermal controls maintained across the full synthesis cycle.
Internal Analysis Protocols
Objectivity is the foundation of our verification. We deploy laboratory-grade testing methodologies to confirm compound purity and structural alignment before release.
Spectroscopy Analysis
Molecular signature verification against baseline structural targets.
Data Yield Compilation
Objective, automated logging of purity metrics for batch consistency.
Global Infrastructure
Direct fulfillment from ZQX-owned facilities. Every shipment originates within our synthesis infrastructure, routed through proprietary logistics nodes engineered for chain-of-custody integrity.

Climate Control
Direct Routing
Chain of Custody

Strict Handling Parameters
Compliance is not variable. All research compounds are subjected to rigid handling parameters to ensure absolute batch consistency and laboratory-grade integrity from synthesis to delivery.
- Zero-tolerance contamination protocols.
- Strict environmental baseline maintenance.
- Standardized laboratory-grade compliance metrics.
- Objective compound stability verification.
Secure Routing
Initiate direct, encrypted communication for high-purity synthesis inquiries and global logistics coordination.